Imaging Facility
Services
High-resolution X-ray Computed Tomography (CT)
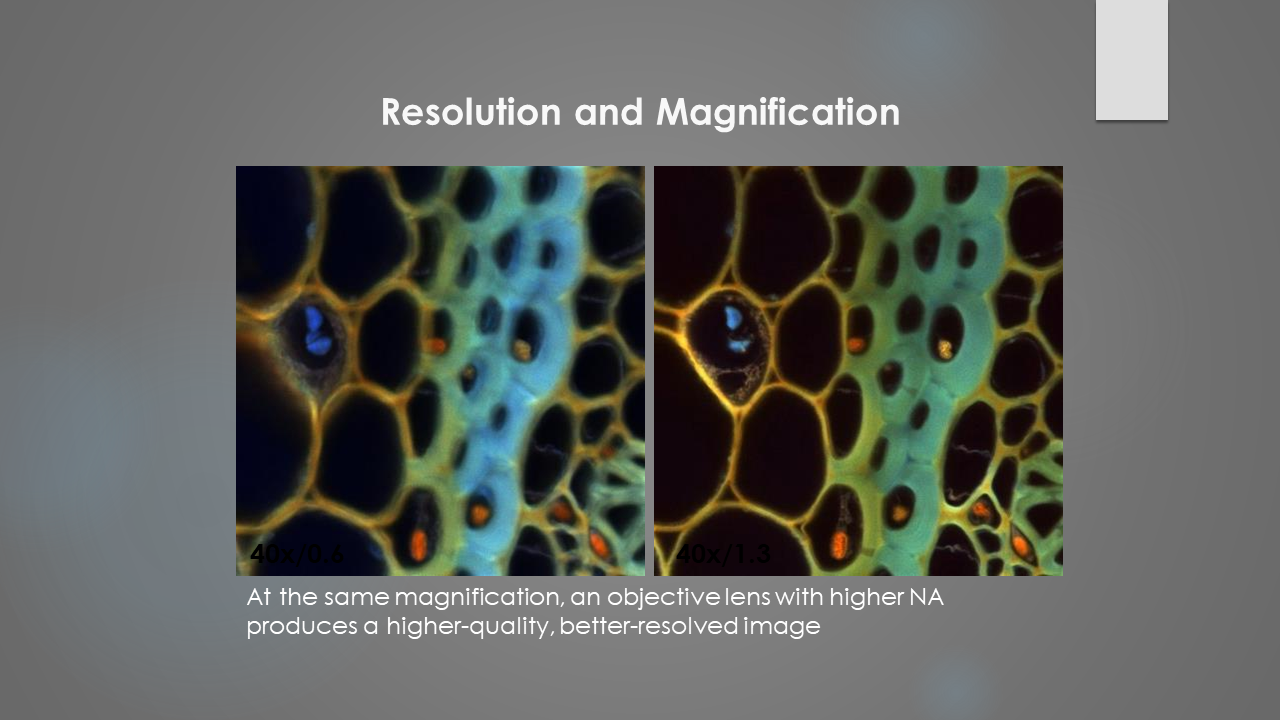
Microscopy
- Super resolution microscope (Zeiss Elyra PS.1)
- Light sheet microscope (LaVision BioTec)
- Confocal multiphoton microscope upright (Zeiss LSM880 “u880”)
- Confocal multiphoton microscope inverted (Zeiss LSM880 “i880”)
- Confocal multiphoton microscope inverted (Zeiss LSM980 "i980")
- Keyence BZ-X810 Microscope
- FVMPE-RS Twin Laser Multiphoton Microscope (Olympus)
- Confocal microscope (Zeiss LSM710)
- Spinning disk confocal microscope (Andor)
- Nikon CSU-W1 spinning disk confocal microscope
- Fluorescence upright microscope (Olympus)
- Fluorescence inverted microscope (Nikon)
- Stereomicroscope (Zeiss)
Cornell Visualization and Imaging Partnership
The Cornell NanoScale Science and Technology Facility (CNF) and the Cornell Institute of Biotechnology (Biotech) have partnered to further advance Cornell’s excellence in Life Science characterization and imaging capabilities through the application of state-of-the art micro - and nanofabrication tools and vice versa. Our shared mission is to foster and enhance the convergence of research fields and bring together ideas and approaches leading to new solutions that will inspire innovation and discovery.
Bioluminescence and fluorescence macro imaging
High resolution ultrasound
Cell Culture Support
Software access
Acknowledging BRC services and staff
If you publish data that has been generated with the help of the services we provide, mentioning the Cornell Institute of Biotechnology in the acknowledgments section is very helpful. Here is why. You can also review our authorship guidelines. Please reference the following Research Resource Identifier (RRID): RRID:SCR_021741